همجوشی هستهای
| فیزیک هستهای | ||||||||

| ||||||||
| واپاشی شکافت هستهای گداخت هستهای واپاشیهای کلاسیک | ||||||||
واپاشی آلفا · واپاشی بتا · پرتوزایی گاما · واپاشی کروی
| ||||||||
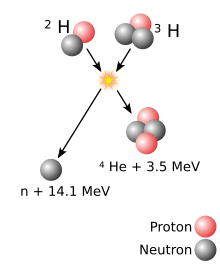
گداخت هستهای،[۱] همجوشی هستهای یا فیوژن (به انگلیسی: Fusion) فرایندی عکس عمل شکافت هستهای است. در فرایند همجوشی هستهای هستههای سبک مانند هیدروژن، دوتریوم و تریتیوم با یکدیگر همجوشی داده شده و هستههای سنگینتر و مقداری انرژی تولید میشود.
برای اینکه همجوشی امکانپذیر باشد هستههایی که در واکنش وارد میشوند باید دارای انرژی جنبشی کافی باشند تا بر میدان الکترواستاتیکی پیرامونشان فائق آیند؛ بنابراین دماهای وابسته به واکنشهای همجوشی فوقالعاده بالاست.
در سال ۱۹۵۲ اولین انفجار آزمایشی گرماهستهای باعث آزاد شدن مقدار زیادی انرژی کنترلنشده شد. این آزمایش نشان داد که اگر دمای یک گاز متشکل از ذرات باردار — پلاسما — با چگالی بالا تا حد ۵۰ میلیون درجه کلوین افزایش یابد، باعث ایجاد واکنش همجوشی هستهای در گاز یونیده میشود. پس از انفجار موفقیتآمیز بمب هیدروژنی جستجو برای آزاد کردن کنترل شده انرژی همجوشی شروع شد.
گرمای همجوشی به مفهوم گرمای حاصله از همجوشی هستهای است.[۲][۳][۴][۵]
همجوشی هستهای، واکنشی کاملاً برعکس شکافت هستهای است. به جای شکافتن اتمهای بزرگ به اتمهای کوچک، اتمهای کوچک به یکدیگر جوش داده میشوند تا اتمهای بزرگ بهوجود آیند. این واکنش انرژی خیلی زیادی آزاد میکند، چرا که طبق نظریهٔ نسبیت خاص انشتین، قسمتی از مادهٔ این واکنش به انرژی تبدیل میشود. واقعیت این است که خارج از نیروگاههای همجوشی و در طبیعت، ما هر روز اثر این واکنش را احساس میکنیم. همجوشی هستهای همان چیزی است که در مرکز خورشید رخ میدهد میکند.[۶]
مزیتها[ویرایش]
مزیت همجوشی هستهای نسبت به شکافت هستهای:
- منابع سوخت آن بسیار فراوان است. به عنوان مثال دوتریوم حدود ۱۵۳ ۰٫۰ درصد اتمی از هیدروژنهای آب اقیانوسها را تشکیل میدهد. تریتیوم نیز در فرایند جذب نوترون توسط لیتیوم قابل تولید است.
- به ازاء هر نوکلئون از ماده سوخت، انرژی تولیدی نسبت به روش شکافت بیشتر است.
- معضل پسماندهای هستهای را ندارد.
- اینکه در هنگام وقوع حوادث احتمالی، رآکتور همجوشی از کنترل خارج نمیشود.
به عنوان مثالی از انرژی تولیدی در یک رآکتور همجوشی میتوان گفت اگر یک گالن از آب دریا را که دارای مقدارکافی دوترون است در واکنش همجوشی استفاده کنیم معادل ۳۰۰ گالن گازوئیل انرژی بدون آلودگی تولید میکند.
سختیهای فرایند همجوشی هستهای[ویرایش]
چیزی که باعث میشود رسیدن به فناوری همجوشی مشکل باشد، عدم علاقهٔ هستهٔ اتمها به جوش خوردن با یکدیگر است. هستهٔ اتم هیدروژن دارای یک پروتون است و بنابراین بار الکتریکی مثبت دارد. وقتی میخواهیم یک هستهٔ اتم هیدروژن دیگر را به آن جوش بدهیم، به دلیل اینکه هر دو دارای بار مثبت هستند، در برابر جوش خوردن مقاومت میکنند. تنها راه این است که به زور این کار را انجام دهیم و آنقدر دمای اتمها را بالا ببریم که به پلاسما تبدیل شوند. اگر پلاسمایی با دمای بسیار بالا داشته باشیم، بعضی از هستهها چنان محکم به یکدیگر برخورد میکنند که به یکدیگر جوش میخورند. برای انجام این فرایند، به دما و فشار خیلی زیادی احتیاج است. مشکل اینجاست که ما بر روی زمین باید شرایط قسمت مرکزی خورشید را بازسازی کنیم. خورشیدی که جرماش ۳۳۰ هزار برابر زمین است و دمای مرکز آن به ۱۷ میلیون درجهٔ سانتیگراد میرسد. مشکل نخست این است که بر روی زمین به اندازهٔ خورشید سوخت هیدروژن در اختیار نداریم، باید دما را به ۱۰۰ میلیون درجهٔ سانتیگراد برسانیم. مشکل دوم که ماده در شکل پلاسما رفتارهای عجیبی از خود نشان میدهد. پلاسما شکل چهارم ماده است نه مایع، نه جامد و نه گاز. وقتی پلاسما را در دما و فشار خیلی زیاد قرار میدهیم، به شدت ناپایدار میشود. برای کنترل شرایط ناپایدار آن نیز از تجهیزات معمولی نمیتوان استفاده کرد. به نوعی باید بر روی زمین یک ستاره ساخت. این کار به قدری چالشبرانگیز است که بشر برای رسیدن به آن، باید پیچیدهترین فناوری تاریخ را بسازد.[۶]
روشهای همجوشی[ویرایش]

محصورسازی به روشهای متفاوتی انجام پذیر است. مهمترین این روشها عبارتند از:
محصور سازی مغناطیسی[ویرایش]
در این روش از میدانهای پرقدرت برای حفظ یک پلاسما استفاده میگردد. سه نوع رآکتور توکامک، اسفرومک و استلراتور بر اساس این روش طراحی شدهاند.
همجوشی هستهای کنترل شده توسط میدان مغناطیسی پر توان[ویرایش]
در این روش از لیزرهای پرتوانی جهت محصورساختن ساچمههای کوچکی استفاده میشود که در آنها سوخت هستهای فشرده سازی شده باشد.
همجوشی هستهای توسط کاتالیزور میونی[ویرایش]
همجوشی هستهای توسط کاتالیزور میونی (Muon-catalyzed fusion (μCF
جستارهای وابسته[ویرایش]
منابع[ویرایش]
- ↑ «گداخت هستهای، گداخت» [فیزیک] همارزِ «fusion»؛ منبع: گروه واژهگزینی. جواد میرشکاری، ویراستار. دفتر اول. فرهنگ واژههای مصوب فرهنگستان. تهران: انتشارات فرهنگستان زبان و ادب فارسی. شابک ۹۶۴-۷۵۳۱-۳۱-۱ (ذیل سرواژهٔ گداخت هستهای)
- ↑ «همجوشی هستهای». بایگانیشده از اصلی در ۲۱ نوامبر ۲۰۱۲. دریافتشده در ۲۹ آوریل ۲۰۱۳.
- ↑ : همجوشی هستهای :
- ↑ «: همجوشی هستهای». بایگانیشده از اصلی در ۱۰ نوامبر ۲۰۱۳. دریافتشده در ۲۵ ژوئیه ۲۰۱۴.
- ↑ Set Cookies
- ↑ ۶٫۰ ۶٫۱ مجله خبر دیجی کالا به نقل از هفتهنامهٔ تایم؛ نسخهٔ دوم نوامبر ۲۰۱۵
- Physics Flexbook Archived 28 December 2011 at the Wayback Machine.. Ck12.org. Retrieved on 2012-12-19.
- Bethe, Hans A. "The Hydrogen Bomb", Bulletin of the Atomic Scientists, April 1950, p. 99.
- The Internal Constitution of the Stars A. S. Eddington The Scientific Monthly Vol. 11, No. 4 (Oct. , 1920), pp. 297–303 JSTOR 6491
- The Most Tightly Bound Nuclei. Hyperphysics.phy-astr.gsu.edu. Retrieved on 2011-08-17.
- What Is The Lawson Criteria, Or How to Make Fusion Power Viable
- "The Multiple Ambipolar Recirculating Beam Line Experiment" Poster presentation, 2011 US-Japan IEC conference, Dr. Alex Klein
- Supplementary methods for "Observation of nuclear fusion driven by a pyroelectric crystal". Main article
- UCLA Crystal Fusion. Rodan.physics.ucla.edu. Retrieved on 2011-08-17. Archived 8 June 2015 at the Wayback Machine.
- Coming in out of the cold: nuclear fusion, for real. Christiansciencemonitor.com (2005-06-06). Retrieved on 2011-08-17.
- Nuclear fusion on the desktop ... really!. MSNBC (2005-04-27). Retrieved on 2011-08-17.
- FusEdWeb | Fusion Education. Fusedweb.pppl.gov (1998-11-09). Retrieved on 2011-08-17.]
- Subsection 4.7.4c. Kayelaby.npl.co.uk. Retrieved on 2012-12-19.
- A momentum and energy balance shows that if the tritium has an energy of ET (and using relative masses of 1, 3, and 4 for the neutron, tritium, and helium) then the energy of the helium can be anything from [(12ET)1/2−(۵×17.6MeV+۲×ET)1/2]2/25 to [(12ET)1/2+(5×17.6MeV+۲×ET)1/2]2/25. For ET=1.01 MeV this gives a range from 1.44 MeV to 6.73 MeV.
- همجوشی هستهای نوشته: مهرداد صمیمی فر؛ انجمن فیزیکدانان جوان ایران
| در ویکیانبار پروندههایی دربارهٔ همجوشی هستهای موجود است. |
