تریاکسید زنون
(تغییرمسیر از زنون تریاکسید)
| تریاکسید زنون | |
|---|---|
| |

| |
xenon trioxide | |
| شناساگرها | |
| شماره ثبت سیایاس | 13776-58-4 |
| کماسپایدر | 21106493 |
| جیمول-تصاویر سه بعدی | Image 1 |
| |
| |
| خصوصیات | |
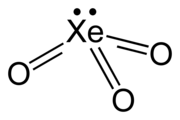
| فرمول مولکولی | XeO3 |
| جرم مولی | 179.288 g/mol |
| شکل ظاهری | colourless crystalline solid |
| چگالی | 4.55 g/cm3 solid |
| دمای ذوب | 25 °C Violent Decomposition |
| انحلالپذیری در آب | Soluble (with reaction) |
| ساختار | |
| شکل مولکولی | trigonal pyramidal (C3v) |
| ترموشیمی | |
آنتالپی استاندارد
تشکیل ΔfH |
402 kJ·mol-1[۱] |
| خطرات | |
| طبقهبندی ئییو | not listed |
| لوزی آتش | |
| ترکیبات مرتبط | |
| ترکیبات مرتبط | تتراکسید زنون اسید زنیک |
| به استثنای جایی که اشاره شدهاست در غیر این صورت، دادهها برای مواد به وضعیت استانداردشان داده شدهاند (در 25 °C (۷۷ °F)، ۱۰۰ kPa) | |
| | |
| Infobox references | |
|
| |
تریاکسیدِ زنون (به انگلیسی: Xenon trioxide) با فرمول شیمیایی XeO۳ یک ترکیب شیمیایی است. که جرم مولی آن ۱۷۹٫۲۸۸ g/mol میباشد. شکل ظاهری این ترکیب، بلورهای دستگاه بلوری چهارگوشه آبی و خاکستری است.این مولکول شکل هرمی دارد.
مولکول این ترکیب درمیدان الکتریکی جهتگیری میکند(قطبی میباشد).
جستارهای وابسته[ویرایش]
منابع[ویرایش]
- ↑ Zumdahl, Steven S. (2009). Chemical Principles 6th Ed. Houghton Mifflin Company. p. A23. ISBN 061894690X.
- «IUPAC GOLD BOOK». دریافتشده در ۱۸ مارس ۲۰۱۲.

